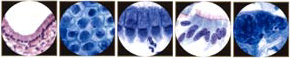
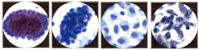
The Pap Smear
Dr. Bruce R. Dziura
Chief, New England Pathology Associates
Springfield, MA (USA)
In Collaboration with Women's Health and Education Center (WHEC)
Before the Pap smear was introduced into clinical practice, carcinoma of the cervix was the leading cause of cancer-related deaths among American women. No other test has been as successful as the Pap smear in eradicating cervical cancer. Cervical cancer is still a leading cause of cancer deaths in women where Pap smear screening is not widely available. Screening for cervical cancer and its precursors with Pap tests represents the most successful cancer detection strategy ever developed. Cervical cytology has remained an extremely effective tool for the eradication of cervical cancer because it takes years or decades for the earliest changes of cervical intraepithelial neoplasia (CIN) to progress to invasive cancer, most cases of CIN regress spontaneously, and most women are screened more than once (1).
This document reflects emerging clinical and scientific advances. This information should not be constructed as dictating an exclusive course of treatment or procedure to be followed. Variation in practice may be warranted based on the needs of the individual patient, resources, and limitations unique to the type of practice or institution.
The American College of Obstetricians and Gynecologists' (ACOG) recommendation on the frequency of Pap test screening are: All women who are or who have been reached age 18 should undergo an annual Pap test and pelvic examination. After a woman has had three or more consecutive, satisfactory annual examinations with normal findings, the Pap test may be performed less frequently in a low-risk woman at the discretion of her physician (2).
Certain high-risk factors have been associated with the development of cervical intraepithelial neoplasia and cervical carcinoma. The ACOG recommends that when one or more of these risk factors is present, more frequent Pap tests may be required. High-risk factors include:
- Women who have had multiple sexual partners or whose male sexual partners have had multiple partners
- Women who began sexual intercourse at an early age
- Women whose male sexual partners have had other sexual partners with cervical cancer
- Women with current or prior human papillomavirus (HPV) infection or condylomata or both
- Women with current or prior herpes simplex virus infections
- Women who are infected with the human immunodeficiency virus (HIV)
- Women with a history of other sexually transmitted diseases
- Women who are immunosuppressed (such as those who have received renal transplant)
- Smokers and abusers of other substances, including alcohol
- Women who have a history of cervical dysplasia or cervical cancer or endometrial, vaginal or valvar cancer
- Women of lower socio-economic status (low socio-economic status appears to be a surrogate for a number of closely related risk factors that place these women at greater risk for cervical cancer)
The cost-effectiveness of cytologic screening for vaginal neoplasia after removal of the cervix for benign disease has not been demonstrated. Nonetheless, periodic cytologic evaluation of the vagina in such cases, based on the above risk factors is warranted.
Methods of Collection:
In the recent past, adaptation in the collection and preparation methods, new methods of interpretation, and the introduction of adjunctive tests have been shown to improve the sensitivity of the Pap test without markedly decreasing its specificity. Originally, cervical cells samples were obtained from the posterior vaginal pool. It has been shown repeatedly that this is a poor source of cells and that the sample should be collected only from the portio and endocervix. Cervical cytology samples should be obtained using either a spatula and endocervical brush or an instrument that can collect cells from both the ectocervix and endocervix.
Slide Preparation:
The Traditional Method: the conventional method of preparation for the laboratory is to spread the collected material on a glass slide and to fix it immediately either by spraying the slide or by immersing it in vial of preservative. To avoid air-drying errors in the interpretation the slide should be fixed right away. Sometimes the smears placed on the glass slide are thick and difficult to analyze.
The ThinPrep: the liquid-based system uses a proprietary fixative into which the clinician places the collection devices. The devices are agitated for a prescribed period, and the vial containing the fixative is sealed, labeled, and forwarded to the laboratory where the slide actually is prepared. The U.S. Food and Drug Administration (FDA) allows the ThinPrep technique to be marketed as better able to detect both low-grade and high-grade squamous intraepithelial lesions (LSIL and HSIL, respectively) than the conventional method of preparation (3).
The SurePath System: it also uses a proprietary preservative solution into which a supplied collection device is placed. The liquid and the collection device are sent to the laboratory. The FDA has approved this technique for use and allows it to be marketed as equivalent to the conventional Pap test. Several other methods of cell collection and preparation currently are in development. None of these had yet received FDA approval.
Automated Slide Interpretation:
Because the interpretation of cervical cytology is labor intensive, there has been great interest in the development of computer-based systems that can read cervical cytology accurately. The FocalPoint slide profiler has been approved by the FDA for primary screening of cervical cytology. This device identifies up to 25% of slides as negative for CIN for which no human review is required (4). Because computerized interpretation may decrease turn-around time and can potentially be cost saving, a number of corporations are developing slide interpretation products. The Bethesda 2001 Terminology Committee suggested that whenever a slide is prepared or interpreted using any form of automation, that fact should be mentioned in the report that is returned to the clinician.
Various Diagnostic Terminologies:
| Traditional Nomenclature | CIN Nomenclature | Bethesda System Nomenclature (SIL) |
|---|---|---|
| Squamous atypia | Squamous atypia | ASCUS |
| Condyloma | Condyloma | Low-grade SIL |
| Mild dysplasia | CIN I | Low-grade SIL |
| Moderate dysplasia | CIN II | High-grade SIL |
| Severe dysplasia | CIN III | High-grade SIL |
| Carcinoma-in-situ (CIS) | CIN III | High-grade SIL |
I. Cytology of Squamous Epithelium:
During normal maturation, the squamous epithelium of the uterine cervix can be conceptualized as differentiating from basal/reserve cells to parabasal cells to intermediate cells to superficial cells. These four cells are the keys to the most common daily diagnostic problem in cytology. The cytoplasm provides information about the origin and functional differentiation of a cell. For this reason, cytoplasmic features are used to determine the degree of squamous differentiation. The cytoplasmic hallmarks of squamous differentiation are distinct cell boundaries and the accumulation of dense cytoplasm. The nucleus provides information about the health of the cell (whether it is normal, inflamed, hyperplastic, or neoplastic). Nuclear features determine where in the continuum of neoplastic transformation, or carcinogenesis, the cell may be. Changes in nuclear size, configuration, and chromatin (hyperchromasia, coarsening, and eventually irregular distribution) and the appearance of visible nucleoli are the main nuclear features of ensuing carcinogenesis.
n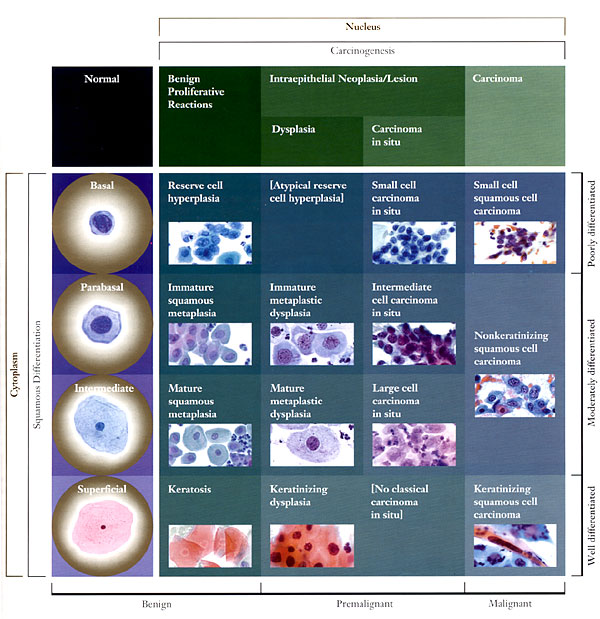
Matrix of Diagnostic Categories
Inflammatory Changes and Specific Infections:
Pruritis and vaginal discharge are among the most common reasons that a woman seeks medical advice from her gynecologist. Although Pap smear can be useful in identifying specific infectious agents, it should not be used in lieu of more effective diagnostic tests. Many things can cause inflammation; the mere presence of inflammatory change on Pap smear is a poor indicator of the presence of infection. Inflammatory change can mimic dysplasia. Patients with persistent inflammatory change are at high-risk for a bona fide squamous abnormality (cervical intraepithelial neoplasia/ squamous intraepithelial lesion).
Differential Diagnosis of Dysplasia vs Inflammatory Change: Inflamed nuclei are big, but not dark; or dark, but not big. Red nuclei indicate inflammation. Dysplastic nuclei are big and dark. Blue nuclei indicate true dysplasia. Dysplasia vs inflammation: matter of degree: more pleomorphic, larger nuclei; more irregular nuclear outline; more abnormal chromatin crisp and distinct; more cellular disorder. Halos: inflammatory vs kilocytotic (5).
| Trichomonas Vaginalis: this is an oval or pear-shaped organism that varies from 8 to 30 micro m. The trichomonad nucleus (thin, elliptical) must be identified to diagnose this infection. Red granules in cytoplasm may be seen. Slightly enlarged, dark nuclei and perinuclear halos are common, mimicking low grade dysplasia. | 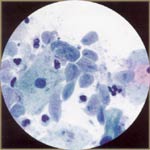 |
| Leptothrix: it is mixed lactobacilli. The organisms are long, thin (less than half as thick as Candida) and flexible. If leptothrix is present, Trichomonas is usually present, but reverse is not true. | 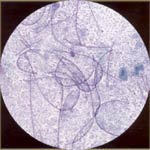 |
| Candida Species: it is associated with a change in vaginal glycogen flora or pH. For example: Pregnancy, late luteal phase of cycle, diabetes mellitus, immunosuppression, debilitating disease, steroids, birth control pills, broad spectrum antibiotics, chemotherapy are associated with candida infection. Pseudohyphae (sticks) and yeast (stones) are seen. | 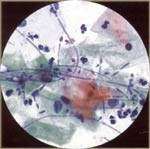 |
| Actinomyces: it is associated with IUD use; rarely is associated with other foreign objects (tampons or pessaries). The patient may be asymptomatic or have pelvic pain. Cytologic findings are colonies of variably gram-positive, long, thin, filamentous bacteria that are reddish, branch are irregularly beaded, and radiate from central area. | 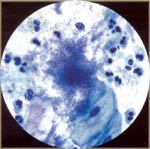 |
| Herpes: it can be asymptomatic or present as blisters, which can ulcerate and be painful. Herpes simplex types I and II are morphologically indistinguishable. The most characteristic cells are multinucleated and the nuclei mold each other. The nuclei are enlarged and the chromatin marginates, resulting in a ground glass appearance. There may be red nuclear inclusions. Late changes are characteristic and diagnostic; early changes can mimic CIN III. Patients with herpes infection are in high-risk group for CIN/SIL. | 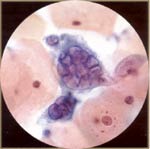 |
| Gardnerella vaginalis (Bacterial Vaginosis): it is a gram-negative, comma-shaped coccobacillus. The bacteria tend to agglomerate onto squamous cells (clue cells). Smears usually show a characteristic granular blue background of small coccobacilli, but the background is otherwise clean, often accompanied by slight parakeratosis. | 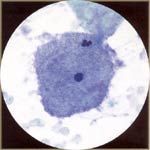 |
| Chlamydia trachomatis: it is an obligate intracellular bacterium that is associated with granular cytoplasmic inclusions. It is the most common cause of non-gonococcal urethritis/ cervicitis. It may cause 20 to 25% of cases of pelvic inflammatory disease, which can result in infertility and ectopic pregnancy. Chlamydia trachomatis is frequently asymptomatic. The value of Pap smear in diagnosing Chlamydia is uncertain. Fine vacuolization of metaplastic cells (having a "moth eaten" appearance) may correlate with high risk of infection. Nebular bodies though rare and difficult to see, may be more specific. Chlamydia changes can mimic low-grade dysplasia. | 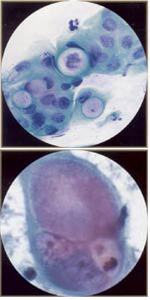 |
II. Cytology of the Glandular Epithelium:
The glandular epithelium of the female genital tract includes the lining of the endocervix, endometrium, and fallopian tube. Pap smear is not nearly as good a screening test for glandular lesions as it is for squamous lesions. Endocervical cells are tall and columnar, and can be secretory or ciliated. Endocervical cells can be seen singly or in strips or sheets. See pictures below: n
Endometrial cells can also form three-dimensional clusters of glandular cells, without central stroma. The cells are small and crowded, and the nuclei are usually degenerated and hyperchromatic can mimic carcinoma in situ. A common everyday problem in Pap smear diagnosis is distinguishing endometrial cells from endocervical cells. This can be important, since shedding of endometrial cells is abnormal in the second half of the menstrual cycle (especially past 40 years of age) or any time in postmenopausal women. Abnormal shedding of endometrial cells carries with it an increased risk of endometrial hyperplasia or neoplasia. See picture below: n
Molecular Screening for Cervical Cancer:
Since the introduction of the Papanicolaou (Pap) test more than 60 years ago, there has been a 70% decline in cervical cancer mortality in the United States, largely attributable to the implementation and widespread acceptance of a cervical cytologic screening program. More than half of the women in the United States who develop cervical cancer have never been screened or have not been screened within the past 5 years. However, cervical cytology has recognized limitations with an estimated sensitivity of only 51%, resulting in the need for repeat cervical cytology at regular intervals to accommodate for its limitation. In the United States, most Pap tests utilize a liquid-based technology as compared with conventional cytology. Persistent infection with oncogenic [high-risk (HR)] human papillomavirus (HPV) has been identified as the underlying cause of cervical cancer (6), leading some to believe that it is time to move from cytologic screening to a molecular screen for HPV DNA. It is highly reproducible, easily monitored, and provides an objective outcome. It is substantially more sensitive than cytology at detecting high-grade cervical intraepithelial neoplasia. However, HPV testing has a lower specificity and is more expensive (7). A lower specificity could result in more downstream testing, such as colposcopy and extirpative procedures, including loop electrocautery excision procedures in patients with only transient infections.
The results of the first screening round of the Canadian Cervical Cancer Screening Trial are recently published (4). The findings from this trial are consistent with previous split-sample and randomized studies, which revealed a higher sensitivity (55% vs 94.6%) for HPV DNA testing as compared with conventional cervical cytology, but a lower specificity. Since this is the first screening round, the authors were not able to address the length of protection afforded by a negative HPV DNA test. Another recently reported study from a Swedish cervical cancer screening program reported a reduction in the incidence of cervical intraepithelial neoplasia 2/3 at subsequent screening in women who were initially screened with HPV DNA and conventional cytology only (8). Their results are consistent with other trials utilizing cervical cytology and HPV DNA testing. Using both tests (HPV DNA and cytology) substantially raised the initial cost of screening. Liquid-based cytology reduces the number of false-negative results as compared with conventional cytology for average risk populations of women, although not for high-risk populations. This technology also reduces the proportion of unsatisfactory specimens compared with conventional smears (3). Based on the potential for improved sensitivity of liquid-based cytology as compared with conventional cytology, the gain in sensitivity for HPV DNA testing may be reduced. A randomized trial of HPV testing and liquid-based cytology in a primary cervical screening involving 25,000 women called A Randomized Trial in Screening to Improve Cytology (ARTISTIC), should address this issue (6).
The current published studies suggest that HPV DNA screening as routine screening is on the horizon. However, large demonstration projects will be needed to evaluate new paradigms for screening, including triage of positive HPV DNA testing with perhaps repeat testing or possibly followed by cervical cytology. The length of protection afforded by a negative HPV DNA test awaits longer term screening data to determine the optimal screening interval. The possibility of self-collected vaginal samples for HPV DNA could further reduce the cost and increase access to screening if rapid, simple, accurate and affordable HPV DNA tests are developed (9). The ultimate goal of cervical cancer screening is to reduce the incidence and mortality from invasive cervical cancer worldwide with a cost-effective, readily available test. We have not yet reached that goal. Potential new paradigms for screening include triage of positive HPV DNA testing with perhaps repeat testing or possibly followed by cervical cytology. The U.S. Food and Drug Administration (FDA) in March 2009 approved the first DNA test that identifies the two types of human papillomavirus (HPV) that cause the majority of cervical cancers among women in the United States (10). The test, called Cervista HPV 16/18, detects the DNA sequences for HPV type 16 and HPV type 18 in cervical cells. Differentiating these HPV types gives health care professionals more information on a patient's risk of subsequently developing cervical cancer. A positive Cervista 16/18 test result indicates whether HPV type 16, 18 or both types are present in the cervical sample. The FDA also approved the Cervista HPV HR test, which is the second DNA test that detects essentially all of the high-risk HPV types in cervical cell samples. The Cervista HPV HR test uses a method similar to the Cervista HPV 16/18 test to detect the DNA sequences of these HPV types. In women age 30 and older or women with borderline cytology, the Cervista HPV 16/18 test can be used together with cytology and the Cervista HPV HR test to assess risk of cervical disease.
Summary:
Although the Pap smear can fail at many levels, ironically, the single most important error is failure of women to get Pap smears in the first place. Cervical carcinoma is preceded by a long prodrome of preinvasive disease that can be detected and treated, which should make it possible to prevent this cancer. The Pap smear is only a screening test for cervical cancer; it has a low, but significant diagnostic error rate. A "negative" report does not guarantee the absence of cervical cancer. Close surveillance of high-risk patients, including those with multiple infections and heavy inflammation, is important. All abnormal Pap smear results should be followed up, and of great importance suspicious lesions should be biopsied and suspicious symptoms investigated, even when the Pap is negative.
References:
- ACOG Technology Assessment in Obstetrics and Gynecology. No 2, December 2002
- Nanda K, McCrory DC, Myers ER et al. Accuracy of the Papanicolaou test in screening for and follow-up of cervical cytologic abnormalities; a systemic review. Ann Int Med 2000;132(10):810-819
- Noorani HZ, Brown A, Skidmore B et al. Liquid-based cytology and human papillomavirus testing in cervical cancer. Ottawa: Canadian Coordinating Office for Health Technology Assessment; 2003. Technology report no 40.
- Mayrand MH, Durato-Franco E. Rodrigues I et al. Human papillomavirus DNA versus Papanicolaou screening tests for cervical cancer. N Engl J Med 2007;357(16):1579-1588
- Rosai J. Uterine cervix - cytology. In: Rosai and Acherman's Surgical Pathology. Publisher: Mosby; ninth edition; 2004
- Kitchener HC, Almonte M, Wheeler P et al. HPV testing in routine cervical screening: cross sectional data from the ARTISTIC trial. Br J Cancer 2006;95(1):56-61
- Koliopoulos G, Arbyn M, Martin-Hirsch P et al. Diagnostic accuracy of human papillomavirus testing in primary cervical screening: a systematic review and meta-analysis of non-randomized studies. Gynecol Oncol 2007;104(1)232-246
- Naucler P, Ryd W, Tornberg s et al. Human papillomavirus and Papanicolaou screening test for cervical cancer. N Engl J Med 2007;357(16):1589-1597
- Wright TCJr, Denny L, Kuhn L et al. HPV DNA testing of self-collected vaginal samples compared with cytologic screening to detect cervical cancer. JAMA 2000;283(1):81-86
- FDA approved first DNA test for two types of human papillomavirus. 2009; http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm149544.htm Accessed 22 October 2009
Published: 2 December 2009
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com