Ultrasound Evaluation of Uterine Scar
WHEC Practice Bulletin and Clinical Management Guidelines for healthcare providers. Educational grant provided by Women's Health and Education Center (WHEC).
Between 1996 and 2003 the cesarean delivery rate in the United States increased dramatically from21.2% to 27.1%. Over the same interval vaginal birth after cesarean birth (VBAC) has decreased from 28.3% to 10.6%. This decrease may be attributed to concerns regarding the risks during trial of labor, such as uterine rupture, estimated to occur in 0.3-4.0% of pregnancies with history of cesarean delivery (1). A trial of labor after previous cesarean delivery has been accepted as a way to reduce the overall cesarean delivery rate. With few exceptions, major improvements in newborn outcomes as a result of the increased cesarean delivery rate are yet to be proved. The dictum "once a cesarean, always a cesarean", which dominated obstetric practice in the United States for nearly 70 years, began changing gradually approximately 30 years ago as improvements in obstetric care made a trial of labor after a previous cesarean delivery safer for both the woman and the fetus. As the VBAC rate increased, so did the number of well-publicized reports of uterine rupture and other complications during trials of labor after previous cesarean deliveries. As a result, many physicians and hospitals have discontinued the practice altogether. This abrupt change in practice has contributed to the cesarean delivery rate in the United States increasing again, reaching an all-time high of 26.1% in 2002, while the VBAC rate has decreased by 55% to 12.6% (2).
The purpose of this document to review the role of ultrasonography in the evaluation of the uterine scar after cesarean delivery. The risk for poor obstetric outcome in a subsequent pregnancy has been shown to be related to surgical technique, with classical cesarean delivery having the highest risk for rupture and lower segment incisions having a lower risk. Clinical significance of uterine scar dehiscence in women with previous cesarean delivery is also discussed.
Prevalence:
Pregnancy after cesarean delivery is a common and major obstetric challenge, with uterine rupture one of its most feared consequences. Four decades ago, due to the catastrophic implications of uterine rupture, contributing to >30% of fetal deaths, vaginal birth after cesarean section (VBAC) was not considered an option. Nevertheless, its practice is still a cause of widespread dispute. Symptomatic or complete uterine rupture involves the entire uterine wall with extrusion of fetal parts and intraamniotic contents in the peritoneal cavity (3). It is accompanied by intraabdominal or vaginal bleeding that requires immediate operative intervention to save the mother and fetus. In contrast, asymptomatic uterine rupture, or uterine scar dehiscence, is usually diagnosed at the time of repeat cesarean delivery. It is defined as separation of the uterine musculature without extravasation of intraamniotic contents and fetal parts into the peritoneal cavity. Usually with dehiscence, the separation does not involve the entire previous uterine scar, the peritoneum overlying the defect is intact, and bleeding is absent or minimal.
The rate of symptomatic rupture of a scarred uterus is about 3.8 per 1,000 trials of labor. The rate of uterine scar dehiscence in the literature is about 13 per 1,000 trial of labor, and it is not significantly different from the rate found during manual uterine exploration after vaginal birth (4). The rate of dehiscence is estimated about 1.9 per 1,000 at the time of repeat, elective cesarean delivery. The fact that the uterine dehiscence is reported to have occurred during repeat, elective cesarean delivery in asymptomatic patients suggests that pregnancy with uterine dehiscence can continue for an indeterminate period. Conventional predictive and diagnostic criteria correlate poorly with the occurrence of scar dehiscence.
Risk Factors:
By using multivariate analysis, several conditions are found as independent risk factors for uterine scar dehiscence. The number of previous cesarean deliveries has the strongest correlation (OR of 2.37). Another risk factor in most of the studies is the rate of non-progress of labor in the first stage. It is well known that contractions in a scarred uterus are a risk factor for the development of uterine rupture, and its association with cephalopelvic disproportion could explain the relationship between failure to progress in the first stage and uterine scar dehiscence. These findings may be explained by the fact that the tissue adjacent to the uterine scar tends to be thinner in patients with 1 or more previous cesarean deliveries and that there is a lack of stretching capability in the scarred tissue (5). Furthermore, during labor, the descent of the fetal head may stretch the lower uterine segment, thereby inducing dehiscence.
It is generally been found that the thicker the uterine scar the lower the rate of complications. One may postulate that a thicker scar is stronger, and thus performs better, than a thinner one. Whether thickness of the uterine scar varies with the technique used at time of hysterotomy closure is unexplored question. One hypothesis is that uterine scar thickness is unaffected by the type of hysterotomy closure. The studies have shown uterine scar thickness diminishes progressively after both one- or two-layer closure but does not vary with mode of hysterotomy closure. The uterine scar thickness remains increased even at 6 weeks post partum, suggesting that the process of uterine scar remodeling extends beyond the traditional postpartum period (6).
Multiparity may have a protective effect against dehiscence in some studies (7). A prior vaginal delivery and VBAC are associated with a higher rate of successful trials of labor as compared with labor in patients with no prior vaginal delivery. Because multiparity is associated with a shorter labor and lower rates of dystocia, it may offer a protective effect against dehiscence due to the lower exposure to contractions and expulsive forces. Another interesting finding is the relationship found between uterine scar dehiscence and preterm delivery (21% of cases as compared with 10.4% of controls, p=0.002). One explanation could be the association between preterm delivery and uterine infection / inflammation. The presence of bacteria in the uterine wall (infection process) may affect the scar tissue, accentuating its part as a locus minor resistentia and making it more susceptible to dehiscence. These results should be taken into consideration whenever a patient with a history of previous cesarean delivery is admitted for preterm labor. In addition, the higher rates of placenta previa could explain the findings as these patients tend to deliver preterm due to vaginal bleeding and the fear of its catastrophic consequences (8).
Labor induction / augmentation: Excessive use of oxytocin may be associated with uterine rupture such that careful labor augmentation should be practiced in women attempting trial-of-labor. Conflicting data also exist whether various induction methods increase the risk for uterine rupture. Uterine rupture is noted in some studies with use of prostaglandin increased the risk 9/366 (2.5%) compared with women induced without prostaglandin use 15/1960 (0.8%). Despite these analyses, it remains unclear whether induction causes uterine rupture or whether an associated risk factor such as cervical status is the ultimate cause. American College of Obstetricians and Gynecologists (ACOG) has issued a committee opinion discouraging the use of prostaglandins for cervical ripening or induction in women attempting VBAC trial-of-labor until this issue is further clarified.
Type and location of uterine scar: Uterine rupture rates are highest with previous classic or T-shaped incisions, with a reported range of 4-9%. The risk for rupture with a previous low vertical incision is difficult to determine. Distinguishing this incision type from classic incision can be arbitrary and low vertical incision is relatively uncommon. It is estimated to be of 0.8-1.1% for prior low vertical scar. Women with unknown scar type may not be at increased risk for uterine rupture. This may simply be because most cases are undocumented prior low transverse incisions. Among 3206 women with unknown scar in the MFMU Network report, uterine rupture occurred in 0.5% of trial-of-labor. Chauhan et al in reviewing 880 maternal uterine ruptures during a 20-year period calculated 40 perinatal deaths in 91,039 trial-of-labor for a rate of 0.4 per 1,000 (9).
Uterine scar evaluation with ultrasonography:
The current methods of diagnosing dehiscence include ultrasonographic evaluation of the lower uterine segment thickness by 2- and 3-dimensional ultrasound; and magnetic resonance imaging; these might be considered in order to enable women to make informed choices regarding VABCs (10). In the immediate postoperative period, it is possible to follow the progression of scar morphology by ultrasonography. In women who have primary cesarean delivery, there is an immediate increase in the thickness of the uterine scar compared with the preoperative myometrial thickness, and then a gradual decrease as the scar is remodeled. It is unclear how long the uterus takes to complete remodeling, and it is possible that significant differences between closure types could appear at longer time intervals. Using magnetic resonance imaging suggests that the remodeling may not be complete up to 6 months after surgery.
These changes in the scar thickness are independent of the method of closure (one- compared with two-layer) and confirmed in some studies. Post operative evaluation of the uterine incision involves identifying the uterine scar in the midsagittal plane perpendicular to the uterine wall by transabdominal or transvaginal approach. The scar is identified by a discontinuity in the architecture of the uterus in the midsagittal plane and is manifest by either a hyperechoic or hypoechoic line perpendicular to the wall of uterus. If transabdominal approach shows poor visualization of the scar, transvaginal ultrasonography is used.
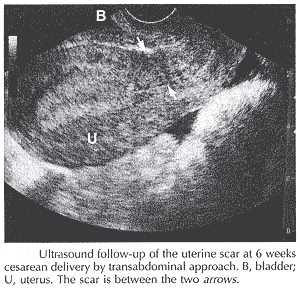
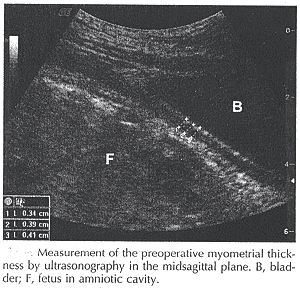
Prior efforts with ultrasound evaluation of the uterine scar have focused on antepartum assessment, and less on postpartum evaluation of the hysterotomy incision repair. Investigators have elucidated the natural history of scar thickness in women with a prior uterine scar and found a correlation between ultrasonographic and clinically determined thickness at cesarean delivery. Some studies have found the antepartum uterine scar thickness inversely correlates with risk of intrapartum rupture, and that intrapartum assessment can predict term intrapartum uterine rupture with a high degree of accuracy (11).
Early diagnosis and management of uterine dehiscence:
Because uterine rupture may be catastrophic, it is recommended that trial-of-labor after prior cesarean delivery should only be attempted in institutions equipped to respond to emergencies, with physicians immediately available to provide emergent care. Thus, an obstetrician and anesthesia personnel must both be available to comply with this recommendations. Recommendations for management of women undergoing a trial-of-labor after prior cesarean delivery are primarily based upon expert opinion. Women attempting VBAC should be encouraged to contact their health care provider promptly when labor or ruptured membranes occur. Continuous electronic fetal heart rate (FHR) monitoring is prudent, although need for intrauterine pressure catheter monitoring is debatable. Studies that have examined FHR patterns prior to uterine rupture consistently report that non-reassuring signs, particularly significant variable decelerations or bradycardia, are the most common finding accompanying uterine rupture.
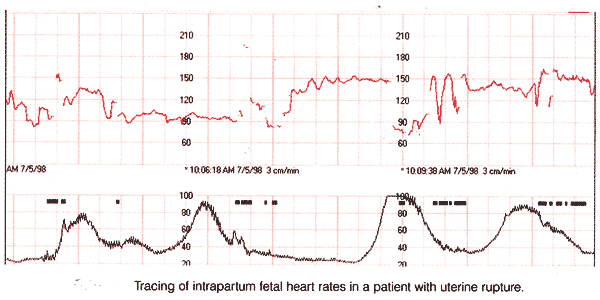
Despite the presence of adequate personnel to proceed with emergency cesarean delivery, prompt intervention does not always prevent fetal neurologic injury or death. If prolonged deceleration is preceded by variable or late decelerations, fetal injury may occur as early as 10 minutes from the onset of the terminal deceleration. Epidural analgesia does not mask the signs and symptoms of uterine rupture. Most individuals do not routinely explore the uterus in order to detect asymptomatic scar dehiscence after VBAC. However, during labor or after delivery excessive vaginal bleeding or maternal hypotension should be promptly evaluated including assessment for possible uterine rupture. Peripartum hysterectomy is usually carried out for life-threatening obstetric hemorrhage and may be regarded as a "near miss" event. "Near-miss" events, defined as " a severe life-threatening obstetric complication necessitating an urgent medical intervention to prevent likely death of the mother" (12). Worldwide maternal deaths from hemorrhage have increased. The cause of this rise is uncertain, but it has been suggested that it may be related to changes in the pattern of childbearing, including an increasing number of cesarean deliveries.
Summary:
In conclusion, it is possible to evaluate, in a longitudinal fashion, the uterine scar by ultrasonography in the postoperative period and during antepartum. Significant variation is seen over time in the scar thickness, and may be unrelated to uterine closure type. Factors that may identify women at risk of uterine scar dehiscence among women attempting VBAC are identified as preterm labor, failure to progress during the first stage of labor and previous cesarean deliveries, post-surgical infection. Multiparity may have a protective effect against dehiscence. Myometrial wound healing knowledge may open novel areas of investigation for potential therapeutic intervention in humans to reduce the risk of rupture during trial of labor after prior cesarean delivery.
References:
- Menacker F. trends in cesarean rates for first births and repeat cesarean rates for low-risk women: United States, 1990-2003. Natl Vital Stat Rep 2005;54:1-8
- Kieser KE, Baskett TF. A 10-year population-based study of uterine rupture. Obstet Gynecol 2002;100:749-753. (Level II-3)
- Ofir K, Sheiner E, Levy A et al. Uterine rupture: Differences between a scarred and an unscarred uterus. Am J Obstet Gynecol 2004;191:425-429
- Cunningham FG, Leveno KJ, Bloom SL et al. Williams Obstetrics. Twenty-second edition. New York, McGraw-Hill, 2005
- Kieser KE, Baskett TF: A 10-year population-based study of uterine rupture. Obstet Gynecol 2002;100:749-753
- Hamar BD, Saber SB, Cackovic M et al. Ultrasound evaluation of the uterine scar after cesarean delivery. Obstet Gynecol 2007;110:808-813
- Hendler I, Bujold E. Effect of prior vaginal delivery or prior vaginal birth after cesarean delivery on obstetric outcomes in women undergoing trial of labor. Obstet Gynecol 2004;104:273-277
- Bashiri A, Burstein E, Rosen S et al. Clinical significance of uterine scar dehiscence in women with previous cesarean section delivery. J Reprod Med 2008;53:8-14
- Chauhan SP, Martin JNJr, Henrichs CE et al. Maternal and perinatal complications with uterine rupture in 142,075 patients who attempted vaginal birth after cesarean delivery: a review of literature. Am J Obstet Gnecol 2003;189:408-417
- Mercer BM, Gilbert S, Landon MB et al. Labor outcomes with increasing number of prior vaginal births after cesarean delivery. Obstet Gynecol 2008;111:285-291
- Asakura H, Nakai A, Ishikawa G et al. Prediction of uterine dehiscence by measuring lower uterine segment thickness prior to the onset of labor: evaluation by transvaginal ultrasonography. J Nippon Med Sch 2000;67:352-356
- Knight M, Kurinczuk JJ, Spark P et al. Cesarean delivery and peripartum hysterectomy. Obstet Gynecol 2008;111:97-105
Published: 5 August 2009
Dedicated to Women's and Children's Well-being and Health Care Worldwide
www.womenshealthsection.com


